Tiziana Life Sciences Ltd. TLSA recently announced a reduction in microglial activation as seen in 3-month Positron Emission Tomography (PET) scans that has now been seen in a total of 5 of the 6 patients with non-active secondary-progressive multiple sclerosis (na-SPMS) treated with intranasal foralumab in its Expanded Access program. Activated microglia are believed to play a prominent role in the pathogenesis of neuroinflammatory diseases including multiple sclerosis, Alzheimer’s disease and amyotrophic lateral sclerosis (ALS).
A reduction in microglial activation is associated with lowered inflammation in the brain. Inflammation in the brain drives the disease pathology in multiple sclerosis. In SPMS, inflammation in the brain occurs in microglia, the brain’s immune cells, which drive the neurodegeneration of brain cells. During the inflammatory process associated with SPMS, microglia are involved in the destruction of myelin, the protective sheath covering of nerve fibers, and contribute to the formation of MS lesions.
Tarun Singhal, M.B.B.S., M.D., Director of PET Imaging Program in Neurologic Diseases, associate neurologist and nuclear medicine physician at Brigham and Women’s Hospital, a founding member of Mass General Brigham Healthcare System, and Assistant Professor of Neurology at Harvard Medical School, commented, “After review of the baseline and 3-month PET scans of the latest cohort of 4 Expanded Access patients, I have determined that 3 out of the 4 patients had a reduction in the microglial PET signal. An example of this can be seen in the graphic below, titled, “Figure 1”, showing the deactivation of this signal in patient EA6. When combined with my assessment of the first 2 Expanded Access patients, a total of 5 out of the 6 had a reduction in qualitative microglial PET signal, which appears to be clearly more significant than what we have identified in our test-retest assessments. I look forward to studying more patients, with full quantitation, and particularly, the next 4 patients in the Expanded Access program to see if this finding is replicated. We are preparing to submit the important results from this trial for publication.”
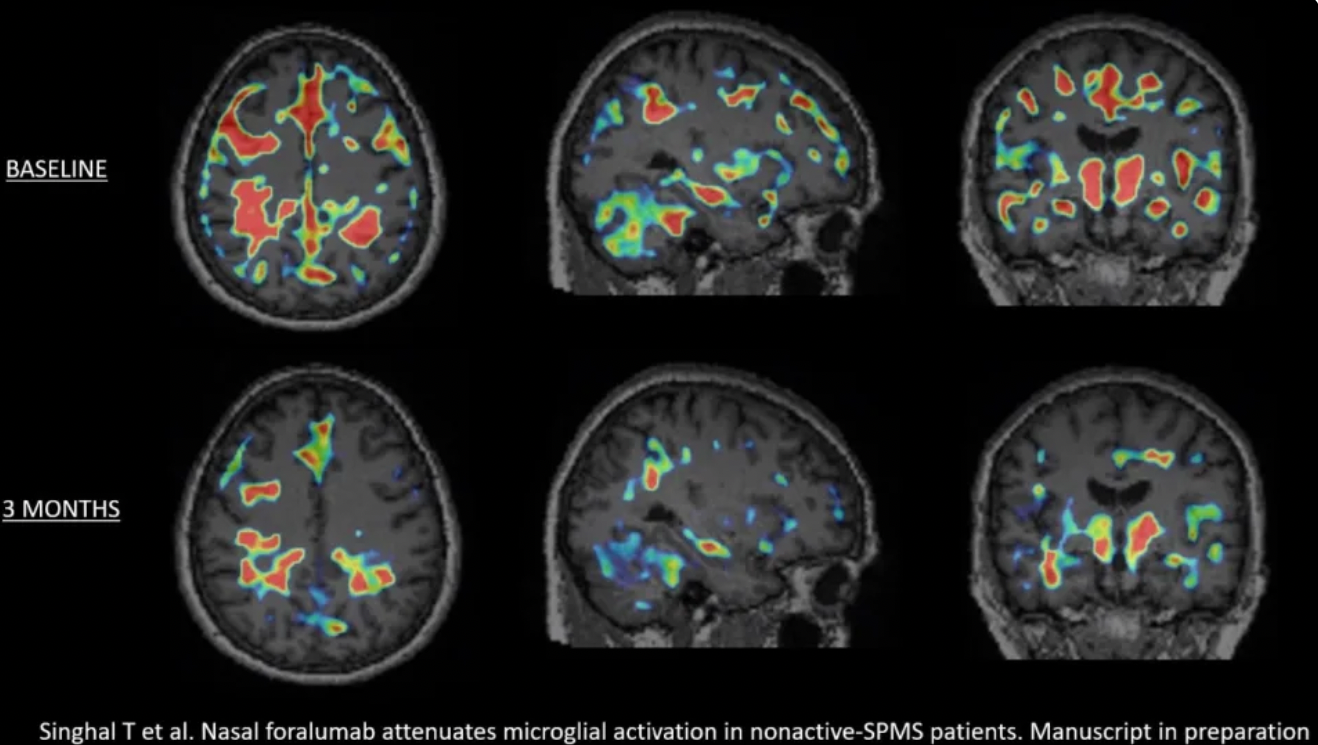
Baseline versus 3-month PET scans
Howard L. Weiner, M.D., Chairman of Tiziana's Scientific Advisory Board and Co-Director of the Ann Romney Center for Neurologic Diseases at Brigham and Women’s Hospital, a founding member of Mass General Brigham Healthcare System, stated, “To see a reduction in microglial activation in 5 out of 6 na-SPMS patients in only 3 months is extraordinary. This finding is even more remarkable because all of the 6 expanded access patients’ na-SPMS had clinically regressed on ocrelizumab treatment. I am excited to follow this program to see if the improvement in the 3-month PET scans will translate to clinical improvement in the coming months.”
Dr. Tanuja Chitnis, M.D., Principal Investigator and Professor of Neurology at Harvard Medical School (HMS) and senior neurologist at Brigham and Women’s Hospital, added, “There are currently no FDA-approved treatments for na-SPMS. The reduction in microglial activation in the 3-month PET scans in 5 out of 6 patients is truly encouraging and I look forward to getting the 3-month PET scans results of the next 4 Expanded Access patients later in 2023 and to starting the Phase 2a trial this year.”
“I believe that Tiziana and Harvard are at the forefront of research in neuroinflammatory diseases with unmet need,” noted Gabriele Cerrone, Executive Chairman, Founder, and acting Chief Executive Officer of Tiziana. “Our Phase 2a multi-center, double-blinded, placebo-controlled trial in na-SPMS uses the 3-month PET scan as the primary outcome measure and our Expanded Access data from the first 6 patients give us increasing conviction in the potential for a positive outcome. We believe this trial design will provide a quick validation of our intranasal foralumab asset and will allow the company to proceed to the next clinical phase of development in na-SPMS.”
Image sourced from Shutterstock
This post contains sponsored advertising content. This content is for informational purposes only and not intended to be investing advice.
Edge Rankings
Price Trend
© 2025 Benzinga.com. Benzinga does not provide investment advice. All rights reserved.
Trade confidently with insights and alerts from analyst ratings, free reports and breaking news that affects the stocks you care about.