AbbVie ABBV announced that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) has adopted a positive opinion, recommending approval of Skyrizi (risankizumab) in the EU to treat adults with moderately to severely active ulcerative colitis (UC).
The eligible patient population includes UC patients who have had an inadequate response, lost response, or were intolerant to either conventional or biologic therapy. The recommended treatment regime with AbbVie's Skyrizi involves an intravenous induction dose of 1200 mg, followed by a subcutaneous maintenance dose of 180 mg or 360 mg, based on individual patient requirements.
Although not legally binding, the EMA will consider the CHMP's recommendation before announcing its final decision, which is expected in the third quarter of 2024.
Please note that Skyrizi is already approved in the United States and EU for treating plaque psoriasis, psoriatic arthritis and Crohn's disease. A potential approval for the UC indication will further expand the drug's label in the EU. UC is a type of chronic inflammatory bowel disease that affects the colon and rectum.
AbbVie has also submitted an application to the FDA, seeking the label expansion for Skyrizi to include the UC indication in the United States. The regulatory filing is yet to be accepted for review.
The stock rose 3.2% in response to the encouraging news. Year to date, shares of AbbVie have gained 4% compared with the industry's 16.3% growth.
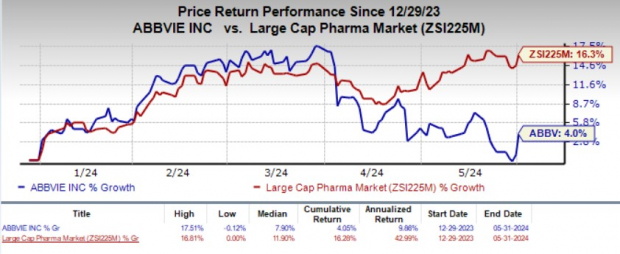
Image Source: Zacks Investment Research
The immunology drug, Skyrizi, is an IL-23 inhibitor that the company has developed in collaboration with Boehringer Ingelheim. AbbVie leads the development and commercialization of the drug globally.
The positive opinion of the CHMP is based on encouraging data from the two phase III studies of Skyrizi (INSPIRE and COMMAND), both of which met the primary endpoint of clinical remission and key secondary endpoints, including endoscopic improvement and histologic-endoscopic mucosal improvement.
Per AbbVie, endoscopic improvement may lead to potential long-term benefits, including lower risk of hospitalizations and improved quality of life for UC patients.
Skyrizi is one of the top contributors to the total revenues generated by the company. In the first quarter of 2024, net revenues recorded from Skyrizi were $2.01 billion, up 48% year over year.
Skyrizi, along with another new immunology drug, Rinvoq (upadacitinib), which is approved for rheumatoid arthritis, ankylosing spondylitis, atopic dermatitis, axial spondyloarthropathy, Crohn's disease and UC, have established outstanding launch trajectories.
Net revenues from Rinvoq in the first quarter were $1.09 billion, up 61.9% year over year.
With approvals for many new indications, the combined sales of these two drugs could be even higher in the future quarters and have the potential to replace blockbuster drug Humira, which has started facing generic erosion in the United States since early 2023. Skyrizi and Rinvoq are expected to collectively exceed the peak revenues achieved by Humira by 2027.
AbbVie expects combined sales (risk-adjusted) of Skyrizi and Rinvoq to be more than $27 billion by 2027 (Skyrizi: more than $17 billion; Rinvoq: more than $10 billion).
Zacks Rank and Stocks to Consider
AbbVie currently carries a Zacks Rank #3 (Hold).
Some better-ranked stocks from the drug/biotech industry are ALX Oncology Holdings ALXO, Annovis Bio ANVS and Entera Bio Ltd. ENTX, each carrying a Zacks Rank #2 (Buy) at present.
In the past 30 days, the Zacks Consensus Estimate for ALX Oncology's 2024 loss per share has narrowed from $3.33 to $2.89. During the same period, the consensus estimate for 2025 loss per share has narrowed from $2.85 to $2.73. Year to date, shares of ALXO have lost 28.6%.
ALX Oncology beat estimates in two of the trailing four quarters and missed twice, delivering an average negative surprise of 8.83%.
In the past 30 days, the Zacks Consensus Estimate for Annovis' 2024 loss per share has narrowed from $2.93 to $2.46. During the same period, the consensus estimate for 2025 loss per share has narrowed from $2.83 to $1.95. Year to date, shares of ANVS have plunged 65%.
ANVS beat estimates in three of the trailing four quarters and missed once, delivering an average negative surprise of 1.39%.
In the past 30 days, the Zacks Consensus Estimate for Entera Bio's 2024 loss per share has remained constant at 25 cents. During the same period, the consensus estimate for 2025 loss per share has remained constant at 54 cents. Year to date, shares of ENTX have skyrocketed 275%.
ENTX's earnings beat estimates in three of the trailing four quarters and missed once, delivering an average surprise of 6.50%.
© 2025 Benzinga.com. Benzinga does not provide investment advice. All rights reserved.
Trade confidently with insights and alerts from analyst ratings, free reports and breaking news that affects the stocks you care about.
