Ultragenyx Pharmaceutical Inc. RARE, along with partner Mereo BioPharma (MREO), announced new positive 14-month data from the phase II portion of the phase II/III Orbit study evaluating setrusumab (UX143) in patients with osteogenesis imperfecta.
The data showed that the large reduction in fracture rates previously reported in patients treated for a minimum of six months was sustained in patients treated for at least 14 months.
OI is a group of genetic disorders that impact bone metabolism. It is characterized by increased bone fragility and a high risk of fractures. Currently, there are no approved treatments for this debilitating condition, representing a highly unmet medical need.
As of the data cut-off date of May 24, 2024, the new data showed that treatment with setrusumab resulted in a large, sustained 67% reduction in annualized fracture rate and persistent median annualized fracture rate of 0.00.
Treatment with setrusumab also resulted in clinically meaningful continued improvement in lumbar spine bone mineral density (BMD) with a mean increase from baseline of 22% at the 12-month timepoint.
Previously, the company reported that treatment with setrusumab for at least six months reduced the annualized fracture rate by 67% in patients with OI.
Per the company, the new data underlines the potential of setrusumab in reducing the incidence of fractures and improving BMD in patients with OI.
No treatment-related serious adverse effects were seen as of the data cut-off date.
Shares of Ultragenyx have plunged 13.4% so far this year compared with the industry's decline of 5.5%.
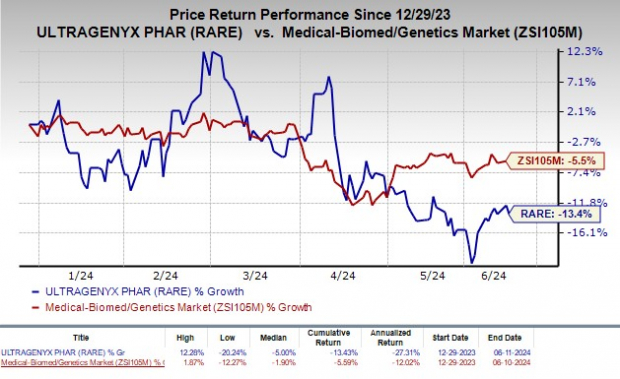
Image Source: Zacks Investment Research
Setrusumab, a fully human monoclonal antibody, is an inhibitor of the sclerostin protein, which is responsible for hampering the maturation and activity of bone-forming cells.
We would like to remind investors that Ultragenyx entered into a licensing and collaboration agreement with Mereo in December 2020 to co-develop setrusumab. Per the terms of the agreement, Ultragenyx leads the global development of setrusumab in both pediatric and adult patients with OI.
As part of this deal, Mereo received an upfront payment of $50 million from RARE, while the latter is eligible for certain regulatory and sales-based milestone payments.
Meanwhile, enrollment in the pivotal phase III portion of the phase II/III Orbit study has been completed. Patients in this portion of the study will be divided into two cohorts in a ratio of 2:1, receiving either a 20 mg/kg dose of setrusumab or a placebo. The primary efficacy endpoint of the study is the annualized clinical fracture rate. Per Ultragenyx, all patients will transition to an extension period and receive open-label setrusumab after completing the primary analysis in the phase III portion of the study.
Apart from the Orbit study, the phase III Cosmic study is also evaluating the effect of setrusumab compared with intravenous bisphosphonate therapy on the annualized total fracture rate in patients aged 2 to <7 years. Enrollment in this study has also been completed.
Zacks Rank & Stocks to Consider
Ultragenyx currently carries a Zacks Rank #3 (Hold).
Some better-ranked stocks in the healthcare sector are Acrivon Therapeutics, Inc. ACRV, Marinus Pharmaceuticals, Inc. MRNS and Minerva Neurosciences, Inc. NERV, each carrying a Zacks Rank #2 (Buy) at present.
In the past 60 days, estimates for Acrivon Therapeutics' 2024 loss per share have narrowed from $3.42 to $2.47. Loss per share estimates for 2025 have narrowed from $3.36 to $2.55. Year to date, shares of ACRV have surged 50.4%.
ACRV's earnings beat estimates in three of the trailing four quarters and missed the same on the remaining one occasion, the average surprise being 3.56%.
In the past 60 days, estimates for Marinus Pharmaceuticals' 2024 loss per share have narrowed from $2.43 to $1.87, while loss per share estimates for 2025 have narrowed from $1.97 to 90 cents. Year to date, shares of MRNS have plunged 85.7%.
MRNS's earnings beat estimates in two of the trailing four quarters, met the same once and missed the same once, the average surprise being 3.27%.
In the past 60 days, estimates for Minerva Neurosciences' 2024 loss per share have narrowed from $3.57 to $1.89. Loss per share estimates for 2025 have narrowed from $4.54 to $3.60. Year to date, shares of NERV have declined 47.1%.
NERV's earnings beat estimates in one of the trailing four quarters while missing the same on the remaining three occasions, the average negative surprise being 54.43%.
© 2025 Benzinga.com. Benzinga does not provide investment advice. All rights reserved.
Trade confidently with insights and alerts from analyst ratings, free reports and breaking news that affects the stocks you care about.