Mesoblast Limited MESO has witnessed a phenomenal run in the year so far. Shares of MESO have risen a whopping 208.2% year to date against the industry's decline of 6.3%.
The massive outperformance can mostly be attributed to positive regulatory updates.
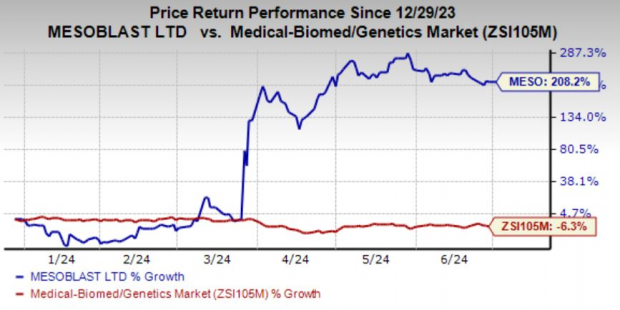
Image Source: Zacks Investment Research
Mesoblast is developing allogeneic (off-the-shelf) cellular medicines for the treatment of severe and life-threatening inflammatory conditions. It is developing product candidates for distinct indications based on its remestemcel-L and rexlemestrocel-L allogeneic stromal cell technology platforms.
Remestemcel-L is being developed for inflammatory diseases in children and adults, including steroid-refractory acute graft versus host disease (SR-aGVHD) and biologic-resistant inflammatory bowel disease. Rexlemestrocel-L is being developed for advanced chronic heart failure and chronic lower back pain.
The company recently confirmed that it will file its biologics license application (BLA), seeking approval of remestemcel-L (brand name Ryoncil) for the treatment of children with SR- aGVHD, with the FDA next week.
Earlier in March 2024, MESO's shares skyrocketed after the FDA informed the company that the available clinical data from the phase III MSB-GVHD001 study of remestemcel-L is adequate to support the resubmission of the BLA for remestemcel-L to treat pediatric patients with SR-aGVHD.
This positive update changed the share price trajectory.
The investors particularly cheered the news as Mesoblast was previously denied marketing approval by the FDA for remestemcel-L twice in the treatment of pediatric SR-aGvHD. The regulatory body had then stated that it required more data to support the approval.
Meanwhile, in March 2024, MESO announced that the FDA supports an accelerated approval pathway for rexlemestrocel-L in patients with end-stage ischemic heart failure with reduced ejection fraction (HFrEF) and a left ventricular assist device.
The agency provided this feedback in formal minutes to the company following the type B meeting held in February 2024, under the existing Regenerative Medicine Advanced Therapy designation.
Rexlemestrocel-L was also granted an orphan drug designation in the United States in February 2024, following the submission of results from the randomized controlled trial in children with hypoplastic left heart syndrome, a potentially life-threatening congenital heart condition. This followed the Rare Pediatric Disease Designation granted by the FDA in January 2024.
These positive updates have boosted investors' hope. A potential approval of Ryoncil should be a significant boost for the company.
We note that Incyte Corporation's INCY lead drug, Jakafi, is approved for the treatment of SR-aGvHD in adult and pediatric patients aged 12 years or older in the United States. Jakafi is a first-in-class JAK1/JAK2 inhibitor.
INCY has a collaboration agreement with the Swiss pharma giant Novartis NVS for Jakafi. Notably, Jakafi is marketed by Incyte in the United States and by Novartis as Jakavi outside the country.
Incyte's Jakafi was initially approved in the United States for the treatment of patients with polycythemia vera (PV), who have had an inadequate response to or are intolerant to hydroxyurea. It is also approved for the treatment of patients with intermediate or high-risk myelofibrosis (MF), including primary MF, post-PV MF and post-essential thrombocythemia MF.
Zacks Rank and a Stock to Consider
Mesoblast currently has a Zacks Rank #3 (Hold).
A better-ranked stock in the biotech industry is ALX Oncology Holdings ALXO, which currently carries a Zacks Rank #2 (Buy).
In the past 30 days, the Zacks Consensus Estimate for ALX Oncology's 2024 loss per share has remained constant at $2.89. During the same period, the consensus estimate for 2025 loss per share has remained constant at $2.73. Year to date, shares of ALXO have plunged 59.5%.
ALX Oncology beat on earnings in two of the trailing four quarters and missed twice, delivering an average negative surprise of 8.83%.
© 2025 Benzinga.com. Benzinga does not provide investment advice. All rights reserved.
Trade confidently with insights and alerts from analyst ratings, free reports and breaking news that affects the stocks you care about.